|
He turned for awhile to the study of the electrolysis of steam, analyzing the appearance of hydrogen and oxygen under various experimental conditions. But by 1892, Thomson was frustrated with difficulties in attaining quantitative measurements in his experiments, and was once again looking for a new approach. The coil produced an electrical field within the bulb that was simpler to study than the one produced by an electrode, and Thomson was optimistic about its results. 
He filled a bulb with gas at a low pressure, and surrounded it with an electrical coil. Thomson refined these investigations by attempting to study the same phenomenon, the discharge of electricity through a gas, with a different experimental arrangement. These experiments, performed with many different gases, provided numerous observations but ultimately led to more puzzles around the relationships among electric current, gaseous discharge, and the chemical combination of atoms. The earliest experiments consisted of passing a discharge between two large parallel plates that functioned as electrodes, in a container filled with gas and connected to a vacuum pump that could vary the pressure within the container. 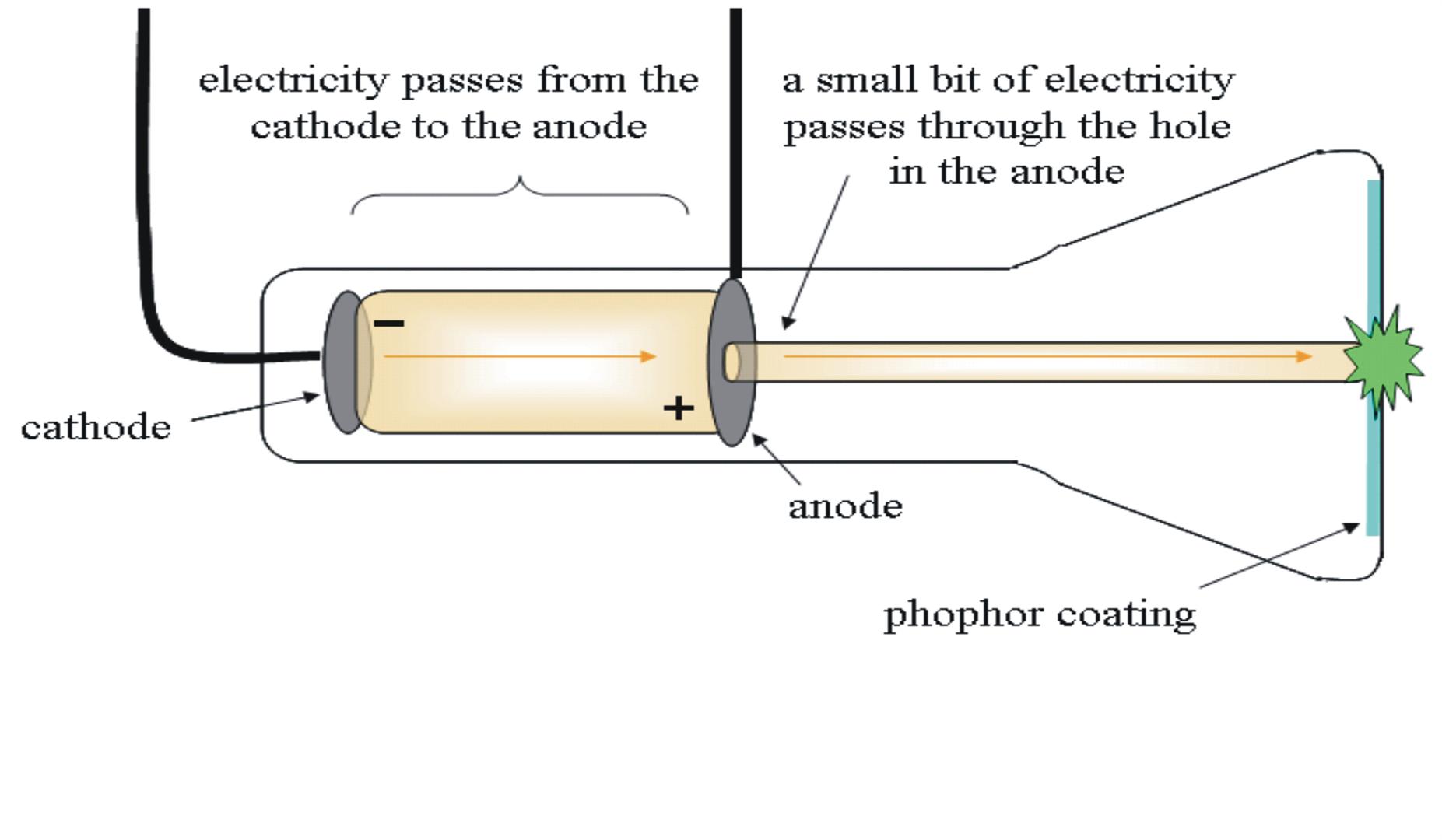
He sought to test various ideas about the dissociation of molecules in an electric field. Soon after taking over his position as head of the Cavendish Laboratory, Thomson began to study the discharge of electricity in gases. Thomson and his colleagues began from Maxwell's framework, but the goals of their research came to include the understanding of fundamental atomic structure and processes. Neither of these methods required an understanding of the nature of the phenomena themselves. In his own work, Maxwell had made use of two important research strategies: he derived mathematical relationships between measurable quantities, and he also created analogies or models between electrical effects and well-understood mechanical devices. This would enable physicists not only to describe the behavior of electricity, but also to understand how its phenomena were produced. An important part of this program was the effort to establish a microscopic basis for electromagnetic phenomena. It remained the preeminent center for the study of subatomic physics into the early decades of the twentieth century.Īlthough Maxwell himself did not set an explicit agenda for his successors, in the period following his death many of Cambridge's physicists devoted themselves to developing, testing, and expanding Maxwell's theory of electricity and magnetism. Mathematical physics had long been established at Cambridge, and while Maxwell, Thomson, and most other Cambridge physicists continued to work successfully in this tradition, the Cavendish Laboratory helped make Cambridge an important center for experimental investigations as well. Although he died in 1879, his influence continued to be felt there among the physicists. Maxwell, who first put forth the theory of an electromagnetic field, set up the Cavendish Laboratory in 1874 as a place to pursue investigations in experimental physics and to provide electrical standards for industry. Joseph John Thomson (1856-1940) spent his professional life at England's Cambridge University, where he passed in four years from prizewinning student (he was ranked second "wrangler" in the prestigious "mathematical tripos" examination in 1880) to head professor at the Cavendish Laboratory-a position previously held by James Clerk Maxwell (1831-1879) and Lord Rayleigh (1842-1919). Along with the nearly contemporaneous discoveries of radioactivity and x rays, the discovery of the electron focused the attention of scientists on the problem of atomic structure, as well as on ways to put these invisible phenomena to use with inventions such as radio and television. Thomson, who in 1897 showed the existence of the charged particles that came to be known as electrons. Both subjects were transformed by the experiments of J. 
Late in the nineteenth century physicists were working hard to understand the properties of electricity and the nature of matter. Thomson, the Discovery of the Electron, and the Study of Atomic Structure Overview
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
